Jurisdiction:
Taiwan
Organ System:
Stomach
Research Organizations:
- Academia Sinica, Taiwan
- National Health Research Institutes, Taiwan
- National Chung Hsing University, Taiwan.
- Kaohsiung Medical University, Taiwan.
- National Taiwan University, Taiwan.
Principal Investigators
:- Yu-Ju Chen
- Ming-Shiang Wu
- Sung-Liang Yu
- Hsuan-Yu Chen
- Deng-Chyang Wu
Publication:
External Links:
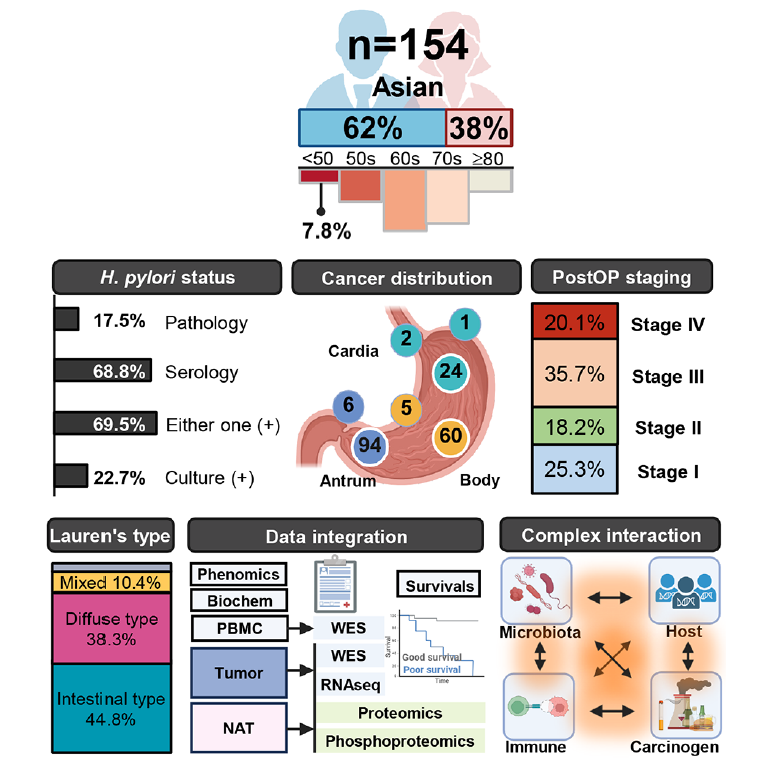
Gastric cancer, with disproportionately higher incidence in East Asia, arises from complex host-microbiome-environment interactions beyond Helicobacter pylori (HP) infection. However, the molecular architecture linking environmental carcinogens, microbial succession and host response remains unclear. To delineate multifactorial aetiologies and clinically actionable subtypes/biomarkers of gastric cancer through integrative proteogenomic, microbial and environmental exposure profiling. We established a multiomics atlas of paired tumour, adjacent mucosa tissues and blood from 154 treatment-naïve Taiwanese patients, integrating whole-exome sequencing, RNA-seq, proteome and phosphoproteome profiling with carcinogen signatures, HP status, microbiome composition and refined anatomical mapping. Cell-based functional assays tested carcinogen effects. Microbial subtype was assessed in an independent cohort. A polycyclic-aromatic-hydrocarbon signature, dibenz[a,h]acridine, emerged as a high-risk exposure promoting invasion, immune suppression and poor survival, significantly exceeding nitrosamine-linked risk in this cohort. Multilayer integration defined three initiation ecologies: HP-driven inflammatory, non-HP microbiome-enriched immune-silent and HP-free microbially depleted states. Among HP-negative tumours, a Streptococcus-enriched subtype associated with tight-junction (CLDN18.2/ZO-1/OCLN) disruption and epithelial-mesenchymal transition, whereas a subset of clinically aggressive cases retained CLDN18.2-high epithelial-stable subtype for therapeutic accessibility. An independent cohort revealed gastric juice-derived Streptococcus anginosus abundance inversely correlated with tight-junction proteins. Anatomical mapping reveals location-specific, sex-specific, subtype-specific oncogenic networks and kinase activity, including CDK4 activation in clinical biomarker-negative tumours. Decision-tree models combining exposure and proteome-immune states refined recurrence and survival prediction beyond stage. This proteogenomic framework defines exposure-informed and microbiome-informed gastric cancer subtypes, providing a molecular schema for patient stratification, prevention and actionable therapeutic vulnerabilities.v